|
While living things have the informational capability to maintain an open system the living things are not subject to law of entropy. For example, heat transfer cannot occur spontaneously from cold to hot, because entropy would decrease. If a locker room is not tidied, entropy dictates that it will become messier and more disorderly over time. Entropy always increases, and rarely ever decreases. 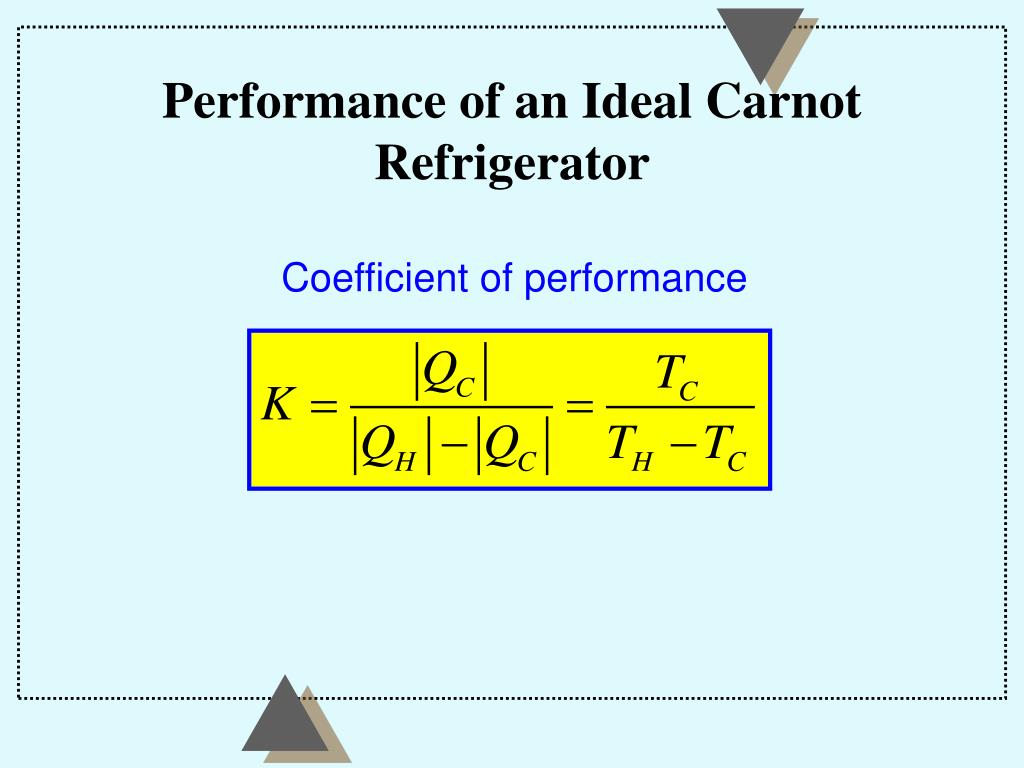
Second law of thermodynamics states that the entropy of any isolated system always increases. When the information is damaged to the point that the organism can not longer overcome the forces of entropy the organism dies.Īll closed systems are subject to the second law of thermodynamics that everything goes from order to disorder. There is a fourth version of the second law of thermodynamics stated in terms of entropy: The total entropy of a system either increases or remains constant in any process it never decreases. This fact balances the first law of thermodynamics. Over time the information in the cell's and organism's DNA is damaged and lost. That is information is lost whenever information is transferred. Shannon's Laws of information show that information transfer is subject to the second law of thermodynamics.  
The energy from outside the system is used to overcome entropy, the spontaneous breakdown of organization within the cells and systems within the living organism. This makes a living organism an open system. Living things die when the disorder in the system of the living organisms increases to the point where the system can no longer function.Ī living organism contains information in the DNA that allows the system to obtain energy from outside the system. The second law says that everything goes from order to disorder, that is an increase in entropy.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
